Their strength indicates that the atoms are difficult to separate, but malleability and ductility suggest that the atoms are relatively easy to move in various directions. 
They are good conductors of heat and electricity. The general properties of metals include malleability and ductility and most are strong and durable. Such bonds are neither ionic nor covalent since the participating electrons are not localized on the atoms. The general observations give rise to a picture of "positive ions in a sea of electrons" to describe metallic bonding. 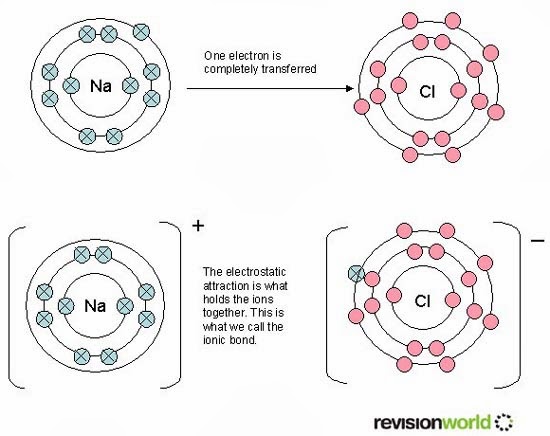
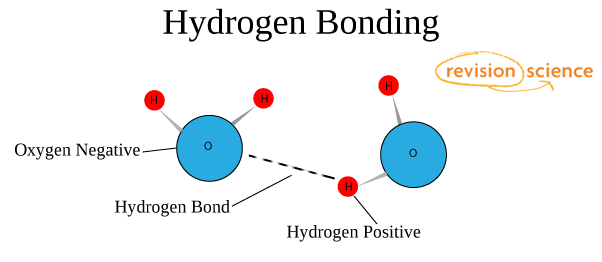
The properties of metals suggest that their atoms possess strong bonds, yet the ease of conduction of heat and electricity suggest that electrons can move freely in all directions in a metal. Ionic bonding can be visualized with the aid of Lewis diagrams. Typical of ionic bonds are those in the alkali halides such as sodium chloride, NaCl. In the extreme case where one or more atoms lose electrons and other atoms gain them in order to produce a noble gas electron configuration, the bond is called an ionic bond. In chemical bonds, atoms can either transfer or share their valence electrons. The ability of an atom to attract electrons in the presense of another atom is a measurable property called electronegativity. In such a bond there is a charge separation with one atom being slightly more positive and the other more negative, i.e., the bond will produce a dipole moment. The nitrogen and oxygen which makes up the bulk of the atmosphere also exhibits covalent bonding in forming diatomic molecules.Ĭovalent bonding can be visualized with the aid of Lewis diagrams.Ĭomparison of ionic and covalent materials.Ĭovalent bonds in which the sharing of the electron pair is unequal, with the electrons spending more time around the more nonmetallic atom, are called polar covalent bonds. The halogens such as chlorine also exist as diatomic gases by forming covalent bonds. Hydrogen gas forms the simplest covalent bond in the diatomic hydrogen molecule. Such bonds lead to stable molecules if they share electrons in such a way as to create a noble gas configuration for each atom. HyperPhysics***** Quantum Physics ***** ChemistryĬovalent chemical bonds involve the sharing of a pair of valence electrons by two atoms, in contrast to the transfer of electrons in ionic bonds. The attractive forces between molecules in a liquid can be characterized as van der Waals bonds. Other types of bonds include metallic bonds and hydrogen bonding. Ionic bond: bond in which one or more electrons from one atom are removed and attached to another atom, resulting in positive and negative ions which attract each other. The two extreme cases of chemical bonds are:Ĭovalent bond: bond in which one or more pairs of electrons are shared by two atoms. The bound state implies a net attractive force between the atoms. A stable compound occurs when the total energy of the combination has lower energy than the separated atoms. Chemical compounds are formed by the joining of two or more atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
